Gomekli is the second ever FDA-approved treatment for rare tumor disease, NF1-PN and the first to be approved for both adult and pediatric patients.

Gomekli is the second ever FDA-approved treatment for rare tumor disease, NF1-PN and the first to be approved for both adult and pediatric patients.

The opinions of Drew Altman, Mark McClellan and Don Berwick
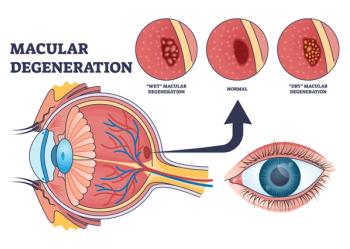
The updated label allows physicians to prescribe Izervay without a limitation on duration for patients with geographic atrophy secondary to age-related macular degeneration.

The group released a statement today expressing that they are eager to work with Robert F. Kennedy Jr. to eradicate HIV when steps into his role as the Secretary of Health and Human Services.

Positive HIV status as well as socioeconomic disadvantages, not breastfeeding, are associated with infant length and birth weight, according to the results of a new South African study.

Those voting to support Robert F. Kennedy Jr as Secretary of HHS include Louisiana Republican Sen. Bill Cassidy. Mitch McConnell, a Republican from Kentucky, however, voted no.

The second part of our conversation with Craig Burton, MBA, executive director of the Biosimilars Council, a trade and lobbying group for the biosimilars industry.

Menopausal patients are most interested in learning more about non-hormonal treatments to address their symptoms, according to the results of an international survey led by the Menopause Priority Setting Partnership (MAPS). Monica Christmas, M.D., director of the menopause program at the University of Chicago Medicine and the Center for Women’s Integrated Health, discussed the survey details with Managed Healthcare Executive.

CVS Health’s healthcare benefits segment, which includes Aetna, had medical benefit ratio of 92.5% in 2024, because of higher utilization, an unfavorable impact of Medicare Advantage star ratings payment and higher levels of care in Medicaid.

We spoke recently with Craig Burton, MBA, executive director of the Biosimilars Council, a trade and lobbying group for the biosimilars industry.
The respiratory tract’s microbial microflora plays a significant role in determining the severity of RSV infections and the development of these long-term conditions.


Judi Health manages medical and pharmacy claims on the same platform to cut administrative cost.

Changing the narrative about health insurance.

The context matters — and it is not a matter of semantics.

Here’s what you missed this week on Managed Healthcare Executive.

The GLP-1 compound drug sold by Hims and Hers is not approved by the FDA and its Super Bowl ad fails to list any risks associated with taking the medication.
Two-thirds of high severity likely due to upcoding, according to a RAND study of hospitalizations in five states.

PEPFAR, the main U.S.-funded global AIDS program was given a waiver from the foreign aid freeze, but there is still uncertainty and interruptions, partly because many PEPFAR programs are implemented through USAID, which has been largely shutdown by the news administration.

Medically tailored meals are an appealing idea, but there is little regulation or standardization.

Lindsay Bealor Greenleaf, head of Federal and State Policy at ADVI Health, talks with Managed Healthcare Executive about Robert F. Kennedy Jr., President Donald Trump’s HHS secretary nominee.

Laurie Ratliff was diagnosed with metastatic breast cancer 25 years ago, and she’s continuing to beat the odds while advocating for aesthetic flat closure as a viable breast reconstruction option.

The technology to leverage healthcare data exists. The problem, according to Highlander Health, is outdated structures and silos.

The quality-adjusted life years has shown staying power in cost-effectiveness calculations, but some say it has major flaws and have proposed alternatives.

An overview of menopause taboo, the lingering effects of the Women’s Health Initiative study and what employers can do support their menopausal workers, according to Stephanie Faubion, M.D., director of the Mayo Clinic’s Center for Women’s Health and medical director of The Menopause Society.
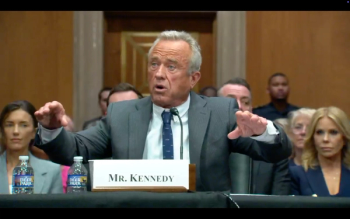
The Senate Finance Committee voted 14-13 along party lines this morning to send the nomination of Robert F. Kennedy Jr. to be HHS Secretary to the full Senate.

The Louisiana Republican, who led a hepatitis B vaccination campaign before entering politics, has expressed concerns about Robert F. Kennedy Jr's views on vaccines.

This follows the company’s recent earnings call, where executives discussed the organization’s growth and future plans, highlighting its focus on serving the best interests of their consumers.

Potential threats to HIV care and fallout over the next four years, according to Jose Abrigo, Esq., HIV project director at Lambda Legal Defense and Education Fund and Carl Schmid, executive director of the HIV+Hepatitis Policy Institute.

Much of the association between trauma and suicide risk among university students can be explained by shame, a new study finds.