
Liraglutide injection is now approved for adult and pediatric patients ages 10 and up, proving another option amid the GLP-1 shortage.

Liraglutide injection is now approved for adult and pediatric patients ages 10 and up, proving another option amid the GLP-1 shortage.

Up to half of obstructive sleep apnea patients taking Zepbound had no symptoms after one year of treatment, averaging 25 fewer breathing interruptions per hour.
If approved, Molbreevi could be the first FDA-approved treatment for patients with autoimmune pulmonary alveolar proteinosis, a rare lung disease.
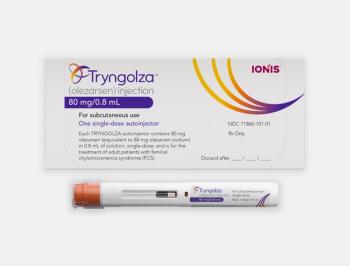
Tryngolza is once-monthly, subcutaneous RNA-targeted therapy and is expected to be available by the end of the year with a price of $595,000.
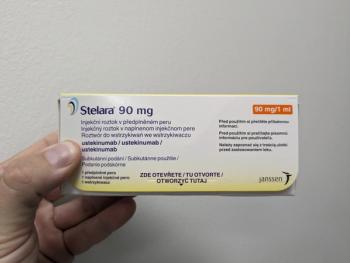
Originally approved in 2009, Stelara patents began expiring in 2023, leading to an influx of recent biosimilars.

If approved, clesrovimab would be the first and only FDA approved single dose immunization for infants approved in time for next year's RSV season, which lasts from October to April.
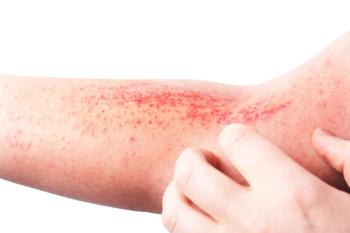
This latest approval is the second indication of the monoclonal antibody, Nemluvio. It was first approved in Aug. 2024 to treat patients with prurigo nodularis.
The combination of Columvi, gemcitabine and oxaliplatin is the first CD20xCD3 bispecific antibody to show positive results in a randomized diffuse large B-cell lymphoma phase 3 trial. The FDA’s decision is expected by July 20, 2025.

The FDA has accepted a new drug application for aficamten, a new obstructive hypertrophic cardiomyopathy drug. A goal date has been set for Sept. 26, 2025.
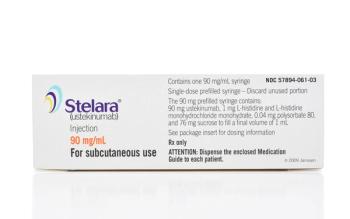
Yesintek is the latest Stelara biosimilar to gain FDA approval and it will be available in February 2025.
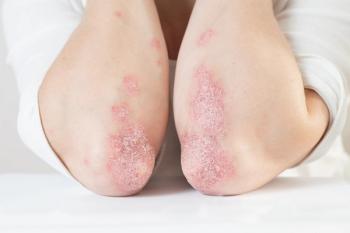
If granted, Tremfya will be approved to treat children ages six and under with severe plaque psoriasis and children ages five and under with juvenile psoriatic arthritis.

Vutrisiran is the generic form of previously approved Amvuttra. The FDA’s target date for the treatment of transthyretin amyloidosis with cardiomyopathy is March 23, 2025.
Imkeldi is a new formulation of imatinib approved as a strawberry-flavored, shelf-stable liquid designed to be more appealing to a wider range of patients, pediatric patients included.

Attruby (acoramidis) is a small molecule approved to treat adults with transthyretin amyloid cardiomyopathy. It will have a list price of $18,759 for a month’s supply.
Produced in Chinese hamster ovary cells, Ziihera (zanidatamab-hrii) is the first HER2-targeted bispecific antibody treatment for patients with previously treated, unresectable or metastatic biliary tract cancer.

After a three-year negotiation, the FDA has dropped its objection to allowing patients to self-titrate dosing of smoked cannabis. But regulators want to see additional information about the device that will be used for inhalation.
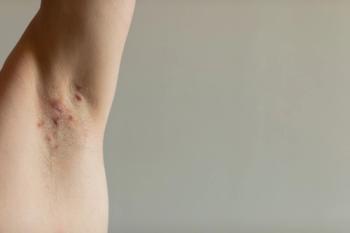
The FDA has approved UCB's Bimzelx for moderate-to-severe hidradenitis suppurativa, offering a new treatment option for this painful autoimmune skin disease.
Relapsed or refractory acute leukemia with a KMT2A translocation currently has an overall survival rate of less than one year when treated with frontline therapies.
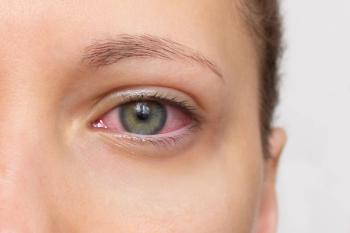
The FDA assigned a Prescription Drug User Fee Act (PDUFA) date of April 2, 2025, for reproxalap for patients with dry eye disease. Reproxalap also is being tested in allergic conjunctivitis.
The FDA has provided a date of April 18, 2025, to review the application for Dupixent to treat patients with chronic spontaneous urticaria, an inflammatory skin disease.
After feedback from the FDA, the companies have voluntarily withdrawn the previous biologics licensing application for datopotamab deruxtecan for patients with advanced or metastatic nonsquamous non-small cell lung cancer.
Aucatzyl is a CAR T-cell therapy that targets CD19 and has been designed to minimize excessive activation of the programmed T cells. The wholesale acquisition cost is $525,000

An FDA advisory committee last year agreed that the data do not support phenylephrine as an effective nasal decongestant.

If approved, donidalorsen would be a first-in-class RNA-targeted medicine for hereditary angioedema. The agency’s goal date is August 21, 2025.

Committee members said there was uncertainty around sotagliflozin in patients with kidney disease. The FDA is currently reviewing the oral therapy as an adjunct to insulin to help control glycemic levels in adults with type 1 diabetes and chronic kidney disease. The agency’s goal date is Dec. 20, 2024.