
Tony Little, ND, VP of Solutions Architecture at Prescryptive Health, shares how the organization is tackling one of the biggest barriers to prescription adherence — cost — through a digital app, among the other barriers patients face.

Tony Little, ND, VP of Solutions Architecture at Prescryptive Health, shares how the organization is tackling one of the biggest barriers to prescription adherence — cost — through a digital app, among the other barriers patients face.

This week, the FDA granted full approval to the Alzheimer’s drug Leqembi, set an action for review of zolbetuximab to treat gastric cancers and rejected Amneal’s new drug application (NDA) Parkinson’s disease treatment.
In this population-based cohort study, researchers compared the risks of incident cardiovascular, neurological, and mental health conditions and rheumatoid arthritis in a year following COVID hospitalization to three different groups: pre-pandemic hospitalization for influenza, pre-pandemic hospitalization for sepsis and hospitalization for sepsis post-pandemic.

Medicare-eligible patients need to be enrolled in registry to get Medicare coverage for the anti-beta amyloid drug that is priced to cost $26,500 a year.


The Magellan Rx Management CEO is replacing Ken Paulus, who has held the top job at Prime since May 2019.

The Magellan Rx Management CEO is replacing Ken Paulus, who held the top job at Prime since May 2019.
7 of them launched during the first several days of July, joining Amgen's Amjevita in the suddenly crowded market of Humira competitors. Pfizer’s Abrilada is the only FDA-approved Humira biosimilar that hasn’t launched.

Walmart has opened 70 Specialty Pharmacies of the Community locations across the U.S. in June, and is expected to open 80 more in 11 states by the end of the year.

Boehringer Ingelheim is pricing its biosimilar 5% to 7% below AbbVie’s top selling drug.

Approvals this week include: a $2.9 million gene therapy for hemophilia A; a pediatric growth hormone; the first cell therapy for type 1 diabetes; a new alopecia drug; a therapy for generalized myasthenia gravis; and full approval for Blincyto in B-cell ALL. The agency also rejected a higher dose of Eylea and set an action for hemophilia B gene therapy.

Stroke patients treated in home-based and hospital-based rehabilitation had mean scores of QOL assessment improved significantly after intervention. Though, the hospital-based group had better improvement than the home-based group in mobility, self-care, pain/ discomfort and depression/anxiety.

Data revealed that just one minute of downtime costs the average business $5,600.

Removing out-of-pocket expenses for the accessible virtual visits may help improve health equity by reducing medical costs for consumers and encourage earlier interventions. In turn, interventions may reduce the risk of complications and avoid visits to more expensive settings like the emergency room.

Data from a survey conducted by Central Michigan University College of Medicine found that newborn weight can be reduced somewhat by marijuana exposure even if it only occurs early in the pregnancy, while reduced head circumference was caused by exposure continued through the second trimester.

Sarah K. Emond, M.P.P., will take over as president and CEO of the Institute for Clinical and Economic Review (ICER) next year. Steve Pearson, M.D.,M.Sc., founded the organization, which has become the leading cost-effectiveness assessment organization of pharmaceuticals in the U.S.

Researchers of a study compared the effectiveness of an automated bilingual computerized alcohol screening and intervention (AB-CASI) digital health tool with standard care for the reduction of alcohol consumption among U.S. adult Latino emergency department patients with unhealthy drinking.

Szczesny, owner and vice president of EDCO Awards & Specialties, gives an in-depth analysis and real-world examples to help steer healthcare organizations in the right direction of value-based operations.
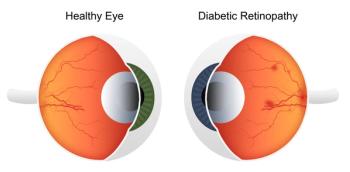
Results reported in JAMA Ophthalmology show that about 20% of those with diabetic retinopathy have a case that could threaten their vision.
This is the first draft guidance accepting considerations in the industry for designing clinical trials for psychedelic drugs.

The FDA’s approvals this week include: a $3.2 million gene therapy, the first anti-inflammatory drug for cardiovascular disease, a subcutaneous version of Vyvgart for myasthenia gravis and Jardiance and Synjardy for kids with type 2 diabetes. Three rejections by the agency include: a therapy for NASH, for retinal cancer, and for rare fungal infections. In addition, Geron has submitted an NDA for first-in-class therapy for MDS.

The Sandoz's and Boehinger Ingelheim's Humira biosimilars will join Amgen's Amjevita on the formulary of UnitedHealth Group's PBM. Cyltezo may have a marketing edge because of its interchangeability designation.

Recently introduced to the U.S. Senate, the Protect Patient Access to Pharmacies Act (S. 2052) will help to secure Medicare patients’, typically seniors, freedom to receive medications and care from the pharmacy of their choice choice through improvements to the “any willing pharmacy” law.

According to a report conducted by the Centers for Medicare and Medicaid Services (CMS) Office of the Actuary (OACT), researchers project healthcare spending growth to pick back up and reach $7.2 trillion by 2031.
Cannabis may accentuate the effects of HIV on the brain or perhaps may mitigate them because of its anti-inflammatory properties.

Patients with multiple sclerosis (MS) with a plant-rich diet had less symptom burden compared with patients with MS who had an elevated intake of red meat and processed meat.

The FDA has approved a second bispecific antibody to treat patients with diffuse large B-cell lymphoma, as well as Linzess to treat children with constipation. Additionally, an FDA advisory committee recommends approval of Sanofi/AstraZeneca’s RSV vaccine for infants.

Improvements have been indicated by a large decrease in the relapse rate during their third trimester of pregnancy.

If passed, PBMs would not be able to charge fees that are connected to the price of a drug, discounts, or rebates.

Virtual care presents a unique opportunity to achieve the kind of whole-person care that has been discussed in healthcare for decades, if not longer.