Following in the footsteps of other pharmaceutical companies, Bristol Myers Squibb and Pfizer are offering Eliquis for cash-paying patients at a discount of 40% off the list price.

Following in the footsteps of other pharmaceutical companies, Bristol Myers Squibb and Pfizer are offering Eliquis for cash-paying patients at a discount of 40% off the list price.

Leqselvi (deuruxolitinib) 8 mg tablets are approved for adults with severe alopecia areata, a disease that causes the immune system to attack hair follicles, leading to partial or complete hair loss on the scalp and body.
Kerendia was originally approved in July 2021 to reduce cardiovascular and kidney complications in patients with chronic kidney disease (CKD) associated with type 2 diabetes. This new approval extends its use to a broader patient population.

Orsini will be the specialty pharmacy partner for Ekterly.
According to Regeneron, the drug is a bispecific antibody that binds to B-cell maturation antigen (BCMA) on cancerous plasma cells and CD3 on T cells, directing the immune system to attack and destroy multiple myeloma cells.

Optum Rx officials said they now have eliminated reauthorization requirements for more than 140 chronic disease medications.

A separate Prime analysis, however, of year-over-year claims data for Wegovy and Zepbound only found an improvement in adherence in 2024, suggesting there is an evolving understanding of obesity treatment.

More than 50 insurers have pledged to streamline and simplify the prior authorization process through six new commitments.
The FDA had resolved the shortage of Wegovy in April, and telehealth providers were advised to stop selling compounded semaglutide products. Novo Nordisk said that Hims & Hers continues to offer these compounded drugs.
This approval marks the eighth disease indication for Dupixent as a new alternative for patients—many of whom are elderly and have limited treatment options.

This is the first FDA-approved CD19- and CD20-targeted immunotherapy combination for adult patients with follicular lymphoma (FL) and the second indication for Monjuvi.

CSL Behring’s Andembry is now approved as a subcutaneous, self-injection for hereditary angioedema (HAE) patients ages 12 and older and will be available by the end of June 2025.
Aldeyra has resubmitted its new drug application for reproxalap, an investigational dry eye treatment, which contained new data from an additional FDA-requested trial.

A second patient with Duchenne muscular dystrophy has died from acute liver failure after receiving the gene therapy Elevidys.

In an interview, James D. Chambers, Ph.D., from Tufts Medical Center, talks about the factors that are impacting the biosimilar market.
The FDA has accepted a New Drug Application for zoliflodacin, a first-in-class oral antibiotic for treating uncomplicated gonorrhea, offering a potential new option amid rising antibiotic resistance.
Ibtrozi had a 90% response rate in treatment naïve adults with locally advanced or metastatic ROS1-positive non-small cell lung cancer, according to the results of a news release.

Enflonsia is the first FDA-approved respiratory syncytial virus (RSV) preventative for infants, regardless of weight, according to the news release.

A survey by PSG has found that the pressure to control drug costs is leading some payers to look into unbundling pharmacy benefits as a way for better control.

Switching to Humira biosimilars saved Navitus clients more than $315 million in upfront costs in 2024 and resulted in a 60% reduction in net costs per claim.

The pharmacy benefit manager (PBM) industry is under pressure to shift to models that avoid conflicts of interest and misaligned incentives.
The approval is based on results from the phase 3 ARANOTE trial, which showed Nubeqa, when combined with androgen deprivation therapy (ADT), reduced the risk of radiographic progression or death by 46% compared to ADT with placebo.

State efforts are aiming to bring more disclosure and transparency to PBM practices, mandating certain pharmacy reimbursement levels, requiring 100% pass-through rebates, and prohibiting spread pricing.
Pharmacy costs and outpatient facility care generate 69% of the cost increase for 2025, according to Milliman’s recent Medical Index.

Jason R. Smith, Pharm.D., appointed as chief pharmacy officer in February 2025 for the University of Rochester Medical Center, talks about building a stronger workforce, managing drug shortages and keeping up with changes in regulations because of the new administration.
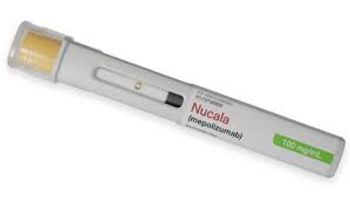
Nucula’s new indication is as an add-on therapy for patients with COPD who also have an elevated blood eosinophil count.