
During an investor call, CVS Health executives said the new legislation requiring changes in PBM business practices was manageable, but they expressed some concern about CMS’s Medicare advanced rate notice.

During an investor call, CVS Health executives said the new legislation requiring changes in PBM business practices was manageable, but they expressed some concern about CMS’s Medicare advanced rate notice.

A recent analysis shows that most products subject to price reduction under the Inflation Reduction Act have achieved substantial returns on investment by the time of negotiations.

PBM reform has finally made its way through Congress and was signed into law by President Donald Trump. Here are seven things you need to know about the law.

Cigna reported 11% revenue growth for 2025. Company executives focused on several key issues in its call with investors: its settlement with the Federal Trade Commission, a transformation announced last year to move to a rebate-free model in pharmacy benefits, and a focus on patient affordability.

Express Scripts agreed to overhaul its business model, ensuring patients pay the lowest available price for medications and reimbursing community pharmacies based on actual costs.

CMS announced 15 drugs for Medicare price negotiations under the IRA, representing $27 billion in spending and treating conditions such as diabetes, cancer, and autoimmune conditions. Here are three insights on the list from industry experts.

During an investor call, UnitedHealth Group executives talk about the steps it has taken to increase transparency, use technology to reshape its healthcare delivery and address how it adapting to financial pressures across its Medicare, Medicaid, and Affordable Care Act businesses.

In a Q&A, Jeffrey Hogan, managing director and co-founder of Judi Group, talks about the importance of identifying hidden costs and contract conflicts of healthcare vendors.

Legislation reforms pharmacy benefit managers by mandating price transparency, eliminating certain compensation models, and providing CMS funding to enforce contract terms.

Lawmakers from both parties grill insurance companies over claims denials, prior authorization delays, and vertical integration.

Judi Group brings together top benefits specialists, ERISA attorneys, actuarial analysts, and technologists to assess risk and protect fiduciary integrity across vendors and solutions.

Medicare’s recently released drug pricing models GLOBE and GUARD use international price benchmarks to generate government rebates but experts say they are unlikely to reduce costs for patients.

Beginning this month, PBMs in California must prioritize health plan interests over their own, and CVS Caremark’s CalPERS contract includes $250 million in performance guarantees.

The PBM association also appointed Brendan Buck chief communications officer. Both Marin and Buck begin Jan. 20, 2026.
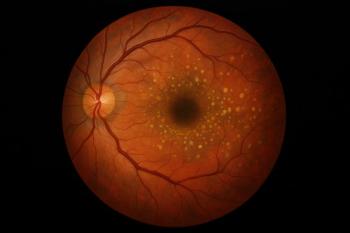
Lytenava, an ophthalmic bevacizumab formulation to treat patients with wet age-related macular degeneration, received yet another FDA complete response letter. The agency is asking for confirmatory efficacy data beyond existing NORSE trial results.

The results of an American Society of Health-System Pharmacists survey shed some light on what is on the minds of pharmacy leaders at health systems.

Drug and hospital prices are top employer healthcare concerns as costs rise, but some employers are unable to access their data, finds a survey by the National Alliance of Healthcare Purchaser Coalitions.
Drug approvals this year have reflected the trend in clinical development of biologics shifting from intravenous to subcutaneous administration, offering shorter injection times, home use, and cost savings for cancer, Alzheimer’s, and inflammatory diseases.

CMS has released the negotiated prices for the second set of drugs for Medicare Part D through the Inflation Reduction Act. Discounts range from 38% for Austedo and Austedo XR to 85% for Janumet and Janumet XR.

Itvisma is a one-time gene therapy approved for both children and adults with spinal muscular atrophy. It will be available in December.

A provision in the One Big Beautiful Bill delays or exempts high-spending medications with orphan indications from Medicare drug price negotiations.

Beginning in January 2026, Optum Rx is removing reauthorizations for two new drug classes: hormone therapies and knee osteoarthritis injectables.
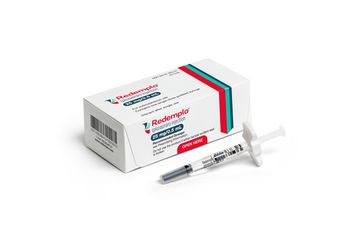
Redemplo has been approved to treat adults with familial chylomicronemia syndrome. The therapy will be available by the end of the year and will have an annual wholesale acquisition cost of $60,000.
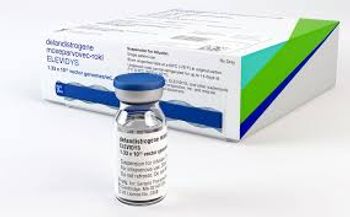
In addition, Elevidys’s indication for non-patients has been removed, and the gene is now indicated only to treat ambulatory patients four years of age and older with Duchenne muscular dystrophy.
Self-injected Brekiya (dihydroergotamine mesylate) is now available by prescription for patients suffering from cluster headaches and migraines with aura.

Foluso Agboola discusses the Institute for Clinical and Economic Review's newest analysis, which has found that most new drugs approved over a three-year period were priced above value benchmarks, potentially costing the healthcare system $1.5 billion.