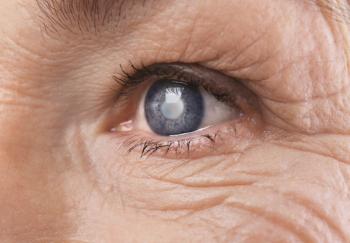
Not Just the Soul. The Eyes Are Also Windows Into Aging
The eye may, indeed, be a window to our souls, but Pankaj Kapahi, Ph.D., is peering into it for other reasons.
His lab the
Kapahi lab researchers conducted a
Existing “
By contrast, the Buck research team based their noninvasive method on evidence that changes in the retina’s microvasculature can warn of age-related diseases such as diabetic retinopathy; neurodegenerative disorders such as Parkinson’s and Alzheimer’s; and systemic diseases including AIDS, chronic hypertension and tumors.
Retinal scans may be more reliable than biomarkers because changes in the eye are less susceptible to day-to-day fluctuations, whereas biomarkers can be influenced by, for instance, meals or infection, Kapahi said. However, subtle changes in the retinal capillaries can often go undetected by even the most sophisticated instruments.
The eyeAge study was conducted in collaboration with Google Research and Zuckerberg San Francisco General Hospital. Using the longitudinal EyePACS dataset, researchers trained a fundus image model on 217,289 examples from 100,692 patients and then tuned it on 54,292 images from 25,238 patients in the UK Biobank. The model showed a strong correlation between chronological age and predicted age in both the EyePACS (0.95) and UK Biobank (0.87) datasets. Notably, eyeAge was also independent of blood marker-based measures of biological age, maintaining an all-cause mortality hazard ratio of 1.026 even when adjusted for phenotypic age.
The researchers also did a genome-wide association study with biological samples from the UK Biobank to establish the genetic basis for the clock. Kenneth Wilson, a postdoc researcher at the Buck Institute, identified nearly 30 genes associated with visual decline, diabetes, hearing loss, Alzheimer’s disease, cardiovascular disease and stroke. One of the genes has been previously shown to extend lifespan in Drosophila (a genus of flies). When Wilson knocked down the gene in the flies, it also improved their vision later in life. “Together, our results suggest that the trajectory of an individual’s biological age can be predicted in timelines under a year and that statistically significant genome-wide associations are possible,” the researchers wrote in their paper, published in eLifeSciences in March.
Because of its ease of use, low cost, and noninvasive sample collection, the retinal aging clock has the unique potential to also assess lifestyle and environmental factors implicated in aging, the researchers say. And the fact that the scans allow for very short timelines means they could be valuable in tracking the efficacy of
“We are looking at aging through a different lens,” Ahadi says, “and bringing more information to the table.”
Newsletter
Get the latest industry news, event updates, and more from Managed healthcare Executive.