Four Ways Treatments for Autoimmune Conditions Could Change
Most autoimmune diseases don’t have cures. Here are the top ways providers can manage and control them.
Most autoimmune diseases don’t have cures. Therefore, physicians try to manage and control them.
“Although many good treatments exist, many patients still suffer with uncontrolled disease and side effects from medications,” says Cynthia Marie Carver DeKlotz, MD, pediatric and adult dermatologist at MedStar Washington Hospital Center and Georgetown University Hospital, and assistant professor of clinical medicine and pediatrics at Georgetown University School of Medicine, all based in Washington, DC. “New and improved treatments are needed.”
Ryan Foster, PharmD, MBA, senior director of pharmacy at Spectrum Health in Grand Rapids, Michigan, says treatment options for many autoimmune conditions are relatively simplistic. “Although advances are being made in personalizing autoimmune treatments, the mechanisms by which current drugs work are limited,” he says. “As our understanding of the underlying etiology of conditions grows, so does our ability to develop targeted therapy options. This will allow providers to create more personalized treatment plans.”
Currently, clinicians choose a particular agent based upon a disease’s clinical features, the patient’s characteristics, such as having other comorbidities and their lifestyle, previous treatment failures, and/or formulary restrictions, says Ronald F. Maceyko, MD, director of the Division of Dermatology at the
Often, the treatment initiated for one type of autoimmune condition can help another one, such as a treatment for psoriasis might also help psoriatic arthritis and inflammatory bowel disease.
Here are four ways treatments for autoimmune conditions could change.
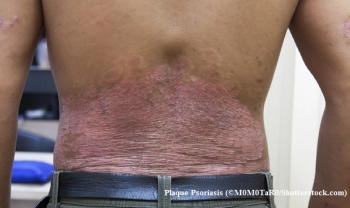
- Biosimilars. These drugs are similar to generic versions of biologic drugs, but because biologics are complex molecules from living sources, they can’t be exactly replicated. Biologic drugs have been emerging as game-changing medications for years in numerous areas of medicine, including autoimmune diseases. “For many patients, biologic medicines can be life changing,” DeKlotz says. “Therefore, once original products' patents expire, it is reasonable to assume that pharmaceutical companies will be interested in developing biosimilar medicines.” Several such biosimilars are already available on the market, including some that treat psoriasis and psoriatic arthritis.
Regarding their use clinically, substantial research and time will tell if a biosimilar proves to be as safe and efficacious as an original biologic medicine. “I believe those factors, in addition to patient access, will ultimately determine if they gain popular use clinically or not,” DeKlotz says.
Recent studies project that biosimilars could reduce spending on biologics between $25 to $150 billion over the next 10 years. This is on top of cost savings from lower-cost generics, which now represent 90% of all medicines given to patients, says Andrew Powaleny, director of public affairs,
- JAK inhibitors. These agents will likely gain significant popularity in the treatment of some, but not all, autoimmune conditions. For example, JAK-inhibitors have been used off label to treat
alopecia areata with success. Currently, according to clinicaltrials.gov, several studies are ongoing to specifically explore their efficacy and safety in treating alopecia areata. Many other autoimmune diseases have trials investigating the safety and efficacy of JAK inhibitors and, in fact, JAK inhibitors are already approved for several autoimmune diseases. “Although they have some serious potential side effects, including serious and potentially life-threatening infections and malignancies, JAK inhibitors can help control certain refractory diseases,” DeKlotz says. “Due to their potentially serious side effects, JAK inhibitors will likely be used with caution. However, as they can be efficacious, their use will likely increase.”
Related:
- Signaling inhibitors. Several interleukin-23 inhibiting drugs have either recently been FDA approved or are in the process of FDA approval. These injectable medications, such as guselkumab and tildrakizumab, combat inflammation and are indicated for use in moderate to severe plaque psoriasis, Foster says.
“Small molecules” as opposed to the very large molecular structure of biologic agents, are a group of newly discovered oral medications that interrupt enzyme signaling pathways between cells that cause inflammation. These include apremilast (Otezla) and tofacitinib (Xeljanz), which have demonstrated efficacy in psoriasis, says Maceyko. Other experimental signaling inhibitors show great promise in treating psoriasis and are anticipated to benefit other autoimmune disorders.
- Laser treatments. Laser treatments are gaining popularity for many diseases, including autoimmune conditions that result in contractures and tightening of the skin. Ablative fractional lasers, such as fractional CO2 lasers, are able to punch micro holes in the skin, relieving the tight contractures in the skin and resulting in increased movement, pliability, and improved range of motion. “This laser treatment may gain popularity in the coming years for select patients with contractures or severe tightening of skin,” DeKlotz says.
“If we are able to find viable treatments that target the root cause of autoimmune conditions, we will be on the path toward a cure,” Foster says. “Although no such therapies are near approval, there are new therapies in research and development.”
Karen Appold is a medical writer in Lehigh Valley, Pennsylvania.
Newsletter
Get the latest industry news, event updates, and more from Managed healthcare Executive.