Cardiac Monitoring at Home Significantly Improves AF Detection
U.S. Preventive Services Task Force hasn't endorsed screening for atrial fibrillation. Findings from these two studies might factor into a reconsideration.
Home-based cardiac monitoring can significantly improve rates of atrial fibrillation (AF) detection, say investigators who completed two recent trials.
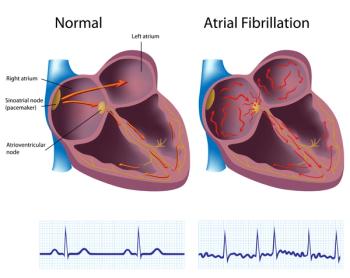
AF is difficult to detect in clinic visits because it may occur only intermittently or be asymptomatic. In the U.S., an estimated 700,000 individuals have undiagnosed AF and half of them are thought to be at moderate to high risk of stroke.
AF also causes — or is associated — with multiple comorbidities, including heart failure, myocardial infarction, chronic kidney disease, thromboembolism and dementia.
However, in January 2022, the U.S. Preventive Services Task Force didn’t take a position for or against screening for AF, citing the lack of evidence about the consequences of screening. These new findings may convince the task force, a group of outside experts that makes advisory recommendations there is a stronger case for screening.
“Although electrocardiograms (ECGs) are readily available in clinics for diagnosis, many patients with AF can present with normal sinus rhythm during the recordings, and thus the diagnostic yield from such one-time testing is poor,” wrote
Noseworthy, a cardioelectrophysiologist in the Department of Cardiovascular Medicine at the Mayo Clinic in Rochester, Minnesota, and a team of investigators determined that equipping patients with wearable cardiac monitors for up to four weeks at a time can improve AF detection rates.
Investigators selected 2,003 patients for wearable monitors and an equal number for the control cohort. Patients were eligible to enroll if they were at risk of stroke and had undergone a 12-lead ECG for any indication but didn’t have known AF.
An AI algorithm was used to stratify patients into high- and low-risk groups based on the ECG data.
The average age of the patients was 74 and almost 40% were women. The average
The trial demonstrated that for high-risk patients, the AI-guided screening significantly improved AF detection over usual care. AF was detected in 10.6% of trial-enrolled high risk patients vs 3.6% of patients in the control group.
The respective percentages for low-risk trial participants and controls were 2.6% and 1.1%. However, the finding in the low-risk trial did not meet standard for statistical significance, so there is some question how large the difference is among peope at low-risk for AF.
This system can help identify high-risk patients and get them into a monitoring program, Noseworthy said “The next step would be to demonstrate that patients who are screening-detected for AF benefit from treatment to prevent a stroke. That would be the biggest motivator to actually screen.”
The
The trial demonstrated that smartphone screening “more than doubled the detection rate of treatment-relevant AF,” wrote lead author Konstantinos D. Rizas, a senior cardiologist at Ludwig Maximilian University of Munich, Germany.
The primary end point for eBRAVE-AF (N = 5,551) was newly diagnosed AF within six months that was treated with an oral anti-coagulant by an independent physician not involved in the study.
According to results reported in Nature Medicine, the end point was reached by 38 patients (1.33%) in the smartphone group compared with 17 (0.63%) in the regular care group during the first half of the trial. In the crossover portion, the respective numbers were 33 (1.38%) vs 12 (0.51%).
The findings were made more relevant because patients were seniors on average and the mean CHA2DS2-VASc score was 3, meaning high stroke risk.
“Intensified digital monitoring using intermittent smartphone PPG [photoplethysmography] measurement is both reasonable and effective to detect AF in a high-risk group,” Rizas and his co-investigators concluded.
Newsletter
Get the latest industry news, event updates, and more from Managed healthcare Executive.