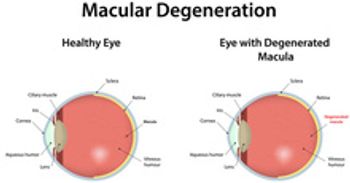
Findings reported in JAMA Network Open today show that Black and Hispanic Medicare beneficiaries with dementia were less likely to use hospice services than White beneficiaries and were more likely to have emergency department visits and hospitalizations.