Researchers found that use of immunosuppressive medication was a strong protective factor, associated with a 60% reduction in mortality.

Researchers found that use of immunosuppressive medication was a strong protective factor, associated with a 60% reduction in mortality.


Research 'marries' traditional pathology with genomics.

The fundraiser is inspired, in part, by Michael Kuchwara, a famed Associated Press theater critic who died from pulmonary fibrosis in 2010

The new chief medical officer of the Pulmonary Fibrosis Foundation speaks about patients in rural areas, research breakthroughs, the drug pipeline, and the Supplemental Oxygen Access Reform (SOAR) Act in an interview with Managed Healthcare Executive.
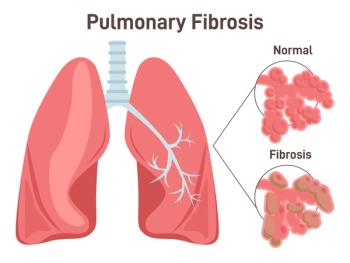
Several weeks after reporting positive results for nerandomilast for idiopathic pulmonary fibrosis, Boehringer Ingelheim reported positive results from a similar phase 3 trial assessing its PDE4B inhibitor as a treatment for progressive pulmonary fibrosis.

Patients with chronic obstructive pulmonary disease, interstitial lung disease and pulmonary hypertension experience improved quality of life and functional capacity as measured in the 6-minute walk distance test.

The biggest challenge in training the next generation of respiratory therapists is getting them to engage in "critical thinking and getting them to think about the disease process," says educator Brandan Gerry. "Students are good at memorizing tidbits but not always assimilating everything into a clear picture and tying things together."
Mayo Clinic researchers find that stem cells with the highest Myc expression become dominant in driving lung remodeling .

The 68-year-old Florida man is walking 106 miles from Key Largo to Key West.

Conventional drug development typically zeroes in on individual proteins or signaling pathways. By focusing on the transcription factor FOXO3, Refoxy hopes to harness a key regulator that impacts numerous biological processes.

Diagnostic criteria for pulmonary fibrosis and other fibrotic diseases in children are lacking. That void hampers an understanding of how disease progresses in children and adolescents and what the outcomes are.

The researchers hope their discoveries will lead to novel strategies to not just slow fibrosis but promote functional repair of the injured lung.
Two industry-sponsored studies investiaged needs of people with pulmonary hypertesion and interstitial lung disease.

Mouse models of liver, kidney and lung fibrosis showed that genetic deletion of MERTK prevented fibrosis
Identifying the underlying causes of prolonged lung inflammation associated with radiographic abnormalities could be key understanding long COVID and other lung conditions, researchers say.
The researchers identified 32 proteins associated with rapid declines in lung function.

The company recently announced promising results from its phase 2a clinical of an agent that the company says is the first to target Traf2- and NCK-interacting kinase, or TNIK for short.

The company said that it will submit a new drug application for the IPF drug, called nerandomilast, based on the positive results.

“Immunity debt” may be the explanation
A nomogram based on four pulmonary factors is helpful but not a substitute for clinical judgment, say the authors.
Pulmonary complications remain a major cause of morbidity and mortality in rheumatoid arthritis-associated interstitial lung disease, despite increasing research interest in the condition.
A new study conducted by researchers at the United Therapeutics Corporation in Durham, NC, looked to characterize PH-ILD disease burden by analyzing healthcare resource utilization (HCRU) and cost data.