In total, 11 lots of three different products have been recalled because of silicone found in retained samples.

In total, 11 lots of three different products have been recalled because of silicone found in retained samples.
Betaxolol may cause a slowing in the heart rate in some patients; an opioid could increase the effect.

Contamination of Brexafemme with a non-antibacterial beta-lactam drug could lead to reactions such as swelling, rash, urticaria and anaphylaxis.

In patients who are immunocompromised, Bacillus cereus can result in life-threatening infections such as endocarditis and necrotizing soft tissue infections.

Crystal formation could lead under- or over-dosing of Sandimmune oral solution, which is used to prevent organ rejection in kidney, liver, and heart allogeneic transplants.

The mix-up in labels can cause either overdosing or underdosing in patients who unknowingly take the wrong dose. Digoxin is used to treat patients with heart failure and chronic atrial fibrillation.

The Association for Accessible Medicines (AAM) published a white paper on the impact that ongoing drug shortages have had on the generic and biosimilar industries, as well as possible solutions stakeholders can put into action.

One of the two recalled lots of the birth control tested high for a known impurity.

The supply of fentanyl, epinephrine injection, and heparin, however, have been impacted by the July 19 tornado at Pfizer's North Carolina facility.

There is a risk the inhaler wouldn’t provide the right dose.
The supply interruption is the result of a combination of factors, including significant increases in demand, an increase in syphilis infection rates, as well as competitive shortages.

Ziprasidone is used to treat patients with schizophrenia and bipolar disorder. Taking dronabinol instead may lead to worsening of mental illness symptoms.

More than 90% of cancer centers have experienced shortages of critical drugs. Erin R. Fox, Pharm.D., University of Utah Health, talks about why these shortages are happening and efforts that are being made to address them.

The FDA has received adverse event reports after patients have used compounded semaglutide that contains salt formulations, which are different active ingredients than that used in the agency-approved drugs for diabetes and weight loss.

The FDA approved Rexulti (brexpiprazole) for Alzheimer’s-related agitation based on the findings of two clinical trials.

One of the warnings regard the risk of opioid-induced hyperalgesia, a condition associated with taking opioids that causes an increase in pain or an increased sensitivity to pain.

Other indications for the blockbuster cancer mediation are not affected.

Issued jointly by the FDA Commissioner and Chief Scientist, the decision means Makena and its generics are no longer approved and cannot lawfully be distributed in interstate commerce. At the same time, the agency “recognizes that there is a supply of product that has already been distributed.”

The presence of a cancer-causing nitrosamine was found in the product, an oral anticoagulant to lower the risk of stroke and blood clots.
Atovaquone Oral Suspension, which treats AIDS-related pneumonia, is being called because of the potential of Bacillus cereus contamination that could lead to life-threatening infections.
Shigella cause an estimated 450,000 infections in the United States each year and an estimated $93 million in direct medical costs.
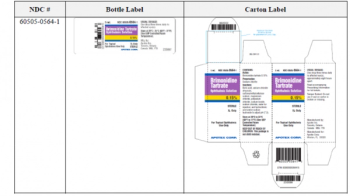
Brimonidine tartrate ophthalmic solution is used to reduce elevated intraocular pressure in patients with open-angle glaucoma or ocular hypertension.

Patients who have had liver problems in the past may be at risk of liver damage from Sprycel, which is used to treat patients with chronic myeloid leukemia.

Hospira/Pfizer’s potassium phosphates product alone may produce daily aluminum exposures of up to twice of the FDA-recommended limit for children.
The products may be ineffective, unsafe and could prevent a person from seeking an appropriate diagnosis.